Clinical Stem Cell Services
GMP iPSC production service for your clinical application
REPROCELL can help with manufacturing your cell therapy product
Clinical Stem Cell Services
REPROCELL can help with manufacturing your cell therapy product
GMP iPSC Master Cell Banks (MCBs) and subsequent cell therapeutics are manufactured by REPROCELL's iPSC experts in compliance with the regulatory standards and guidelines of the FDA, EMA, and PMDA.
Our operational team will provide the necessary quality and regulatory documents such as donor eligibility, CoA, batch records, traceability documentation, and quality technical agreement for your cell therapy project.

Because we have global access to human tissue samples, we procure the tissues needed for your cell therapy project and perform the necessary viral and donor profile screenings ensuring donors are consented for clinical and commercial use. Our experts use our proprietary footprint-free RNA reprogramming technology to generate a StemRNA™ Clinical iPSC Seed Clone Bank from these tissue samples using GMP-grade media and reagents.
Under strict quality control measures, a clinical grade iPSC seed clone can be expanded in a GMP environment to manufacture an MCB which can be further downstream processed to generate a therapeutic cell product.
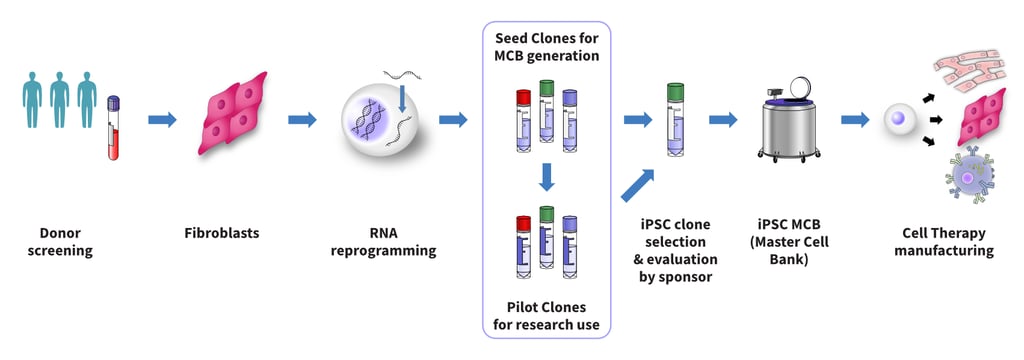
REPROCELL's process starts with collecting skin for fibroblast isolation from screened donors who gave consent for clinical and commercial use, further reprogramming the fibroblasts to iPSCs using our proprietary StemRNA™ 4th Gen Technology. Multiple iPSC clones are isolated and quality controlled to create StemRNA Clinical iPSC Seed Clones, which are suitable for expansion into a GMP iPSC Master Cell Bank.
These clones can be accessed in three ways:
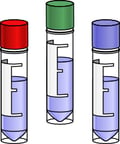
These cells are suitable for clinical use through subsequent regulated and approved processes, including processes resulting in a GMP Master Cell Bank and Working Cell Bank. StemRNA Clinical iPSC Seed Clones are generated using a CGTP process and covered by a rigorous quality control (QC) process that is compliant with US FDA, European EMA, and Japanese PMDA regulations.
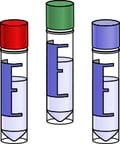
Our non-exclusive, ready-to-use iPSC Pilot Clones are directly expanded from our StemRNA Clinical iPSC Seed Clones in a research setting. These Pilot iPSC Clones are NOT suitable for development for clinical use but are for evaluation purposes only. They provide a more cost-effective way to access StemRNA Clinical iPSCs to develop and evaluate your process.
Both the Seed Clones and the Pilot Clones include an evaluation period to determine their suitability for your process. During the evaluation period, sufficient vials of the Seed Clones will be reserved for you to carry forth into GMP iPSC Master Cell Bank generation after successful evaluation.
|
Donor |
Gender |
Age |
Race |
Clinical Status |
Blood Type |
|
Donor A |
Female |
25 |
Asian |
Healthy |
B+ |
|
Donor B |
Female |
23 |
Caucasian |
Healthy |
A+ |
|
Donor C |
Female |
22 |
Caucasian |
Healthy |
0+ |
|
Donor D |
Male |
23 |
Caucasian |
Healthy |
A+ |
|
Donor E |
Male |
27 |
Caucasian |
Healthy |
A+ |
If you want to find out more about our clinical stem cell services, please contact us.
REPROCELL is a pioneer in using mRNA for generating iPSCs. mRNA generates high-quality iPSCs that are uniquely suited for clinical applications.
To verify the quality of each iPSC line, we perform a multi-level suite of Quality Control assays in line with the regulatory standards. Examples of a few of our assays are shown below.
Morphology, P6+3
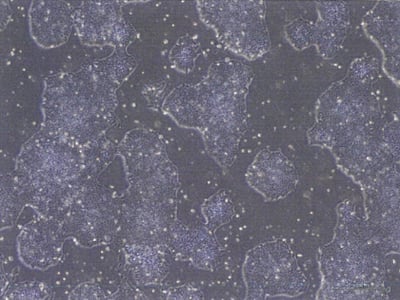
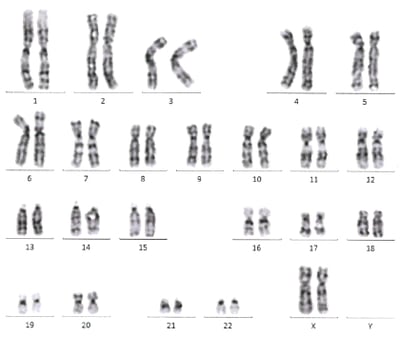
Karyology
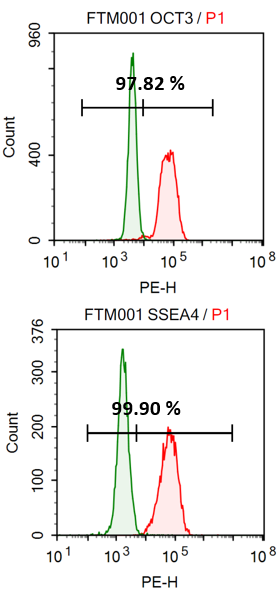
Flow cytometry analysis
3 germ layers differentiation potential
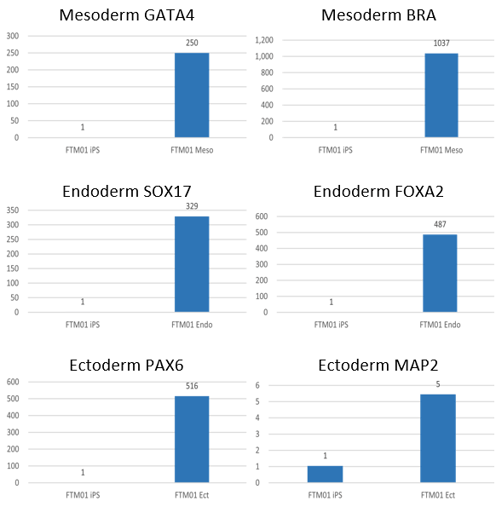
Typical quality control assays of Clinical StemRNA iPSC Lines. To verify the suitability of the StemRNA iPSCs as a starting material for clinical programs each clonal line is validated in several assays, including colony morphology and growth rate, karyotyping, pluripotency marker expression by flow cytometry, and directed differentiation to measure their functional pluripotency potential. Only the clones that pass all of these tests are suitable for advancement to scale-up for GMP Master Cell Bank manufacture.
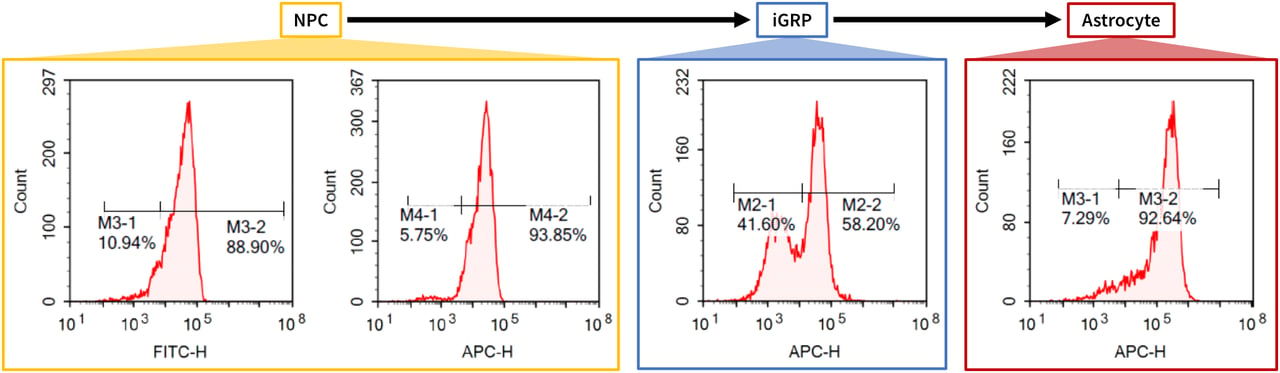
Targeted Differentiation Capacity into Neural Lineage of a Typical Clone. Clinical iPSCs are sequentially differentiated into cells of the neural lineage and characterized by fluorescence cytometry. These results show that at various stages of differentiation, the cells express markers typical for Neural Progenitor Cells, iGRPs (induced glia restricted progenitor cells), and astrocytes. (Data acquired in-house.)
You can also outsource your clinical gene editing project to REPROCELL by providing your own cells or using our StemRNA Clinical iPSC Seed Clones.
Our clients often have concerns about donor consent; but all our tissue donors have fully consented to clinical and commercial use of their cells.
We can also provide the necessary clinical and commercial licenses for your project – making us a hassle-free one-stop solution provider for your iPSC needs.
Also in the following video, REPROCELL’s Chief Operating Officer, Daisuke Usui, summarizes the key benefits of our GMP iPSC Master Cell Bank manufacturing service.
Mr Usui describes the key milestones achieved in each project, the regulatory standards adhered to, and highlights the benefits of choosing REPROCELL over other providers.
At REPROCELL, our scientists understand that your custom iPSC project must be as unique as your research. If you have any questions about how our Clinical iPSC Generation Service can help you advance your project, please make an inquiry using the form below.
Discover more
Resources
Gene Editing Services
Latest in Clinical Capabilities
The REPROCELL Blog
Data Privacy and Protection in AI for Precision Medicine
31 October 2023
Bias in AI for precision medicine
02 October 2023
Clinical Trials: An Evolutionary Perspective
18 January 2023
