Tissue Sourcing for the Development of iPSC-derived Disease Models
Human induced pluripotent stem cells (iPSCs)-based models are an ideal resource for studying disease mechanisms in vitro at the cellular level [1], enabling research studies to be performed under defined experimental conditions and in a reproducible manner.
REPROCELL offers a complete portfolio of services for the reprogramming and differentiation of high-quality iPSCs.
Challenges in creating iPSC-derived models
iPSC-based disease models typically involve multiple steps: obtaining primary target cells, generating iPSCs, differentiating the iPSCs into the disease cell type and culturing the differentiated cells under specialized conditions to develop the disease phenotype in vitro (Figure 1).
One of the main challenges in developing iPSC-based disease models is obtaining the appropriate human tissue to initiate the process. Tissues from individuals with a particular disease allow access to cellular changes that are found in that disease state, and include all of the genotypic changes that underlie a disorder. Cells used in the in vitro model should reflect the genotype and phenotype of the diseased cells in vivo which can be challenging depending on the disorder.
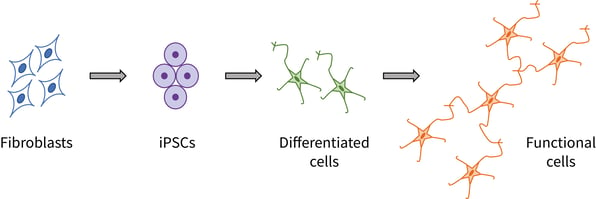
Figure 1. Typical pathway for developing iPSC-based disease models.
How can I access diseased tissues and generate an iPSC-based disease model?
The most straight-forward way to obtain relevant cells to initiate a disease model is to target patients with the disease of interest and obtain a source material, such as blood, urine or skin, from which to isolate primary cells to reprogram (Figure 1). This approach is advantageous in that all of the genetic changes associated with a disease phenotype are incorporated within the primary cell and will therefore be included in the derived disease model. However, disease-affected tissue can be often be difficult to obtain, especially for rarer conditions and disorders.
How can REPROCELL help me to achieve this?
Through our extensive global Tissue Network , REPROCELL can source a variety of diseased and healthy tissue samples (for example skin, blood or urine cells) for use with REPROCELL reprogramming technologies to generate iPSCs on behalf of our clients. Tissues and cells that we source are fully anonymized and consented for use in all types of iPSC-based projects, and come with valuable donor demographic information. We can also perform molecular characterization of tissue or primary cells to provide further genetic information or confirm disease-relevant mutations.
Table 1 shows a few examples of disease areas available through our Tissue Network. Following recovery of tissue, we can source and ship directly to you, isolate the primary target cells and deliver to you, or we can directly carry the primary cells into our reprogramming protocol using our state-of-the-art StemRNA 3rd Gen Reprogramming technology and deliver donor-derived iPSC ready for use (Figure 2). As our network is constantly expanding, please check with us to find out if we can source your target cell or tissue.
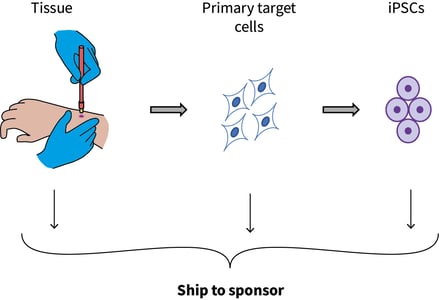
Figure 2. Cells can be shipped at multiple stages
I want to find out more
You can find out more about the REPROCELL Tissue Network, Reprogramming and Molecular Services, using the resources below.
- Global Biorepository of Human Tissue Samples
- iPSC Reprogramming
- Molecular services (Genomic Services)
If you are ready to discuss your research needs with one of our iPSC experts, please please contact us.
| Disease Group | Example Diseases |
| Retinal Eye Disease |
|
| Metabolic Disorders |
|
| Neurological Disorders and Dystrophies |
|
Table 1: Examples of Tissue Network Disease Area

REPROCELL offers a complete portfolio of services for the reprogramming and differentiation of high-quality iPSCs.
References
-
Sarkar et al. Efficient generation of CA3 neurons from human pluripotent stem cells enables modeling of hippocampal connectivity in vitro. Cell Stem Cell 22:684 (2018)
-
Czerniecki et al. High-throughput screening enhances kidney organoid differentiation from human pluripotent stem cells and enables automated multidimensional phenotyping. Cell Stem Cell 22:929 (2018)
-
Holmgren et al. Expression profiling of human pluripotent stem cell-derived cardiomyocytes exposed to doxorubicin – Integration and visualization of multi-omics data. Tox Sci 163: 182 (2018)

Author
Sarah Eminli-Meissner, PhD
Head of Business at REPROCELL
Previously Global Director at Stemgent, Sarah is now responsible for driving REPROCELL's stem cell business revenue. You can contact her on
LinkedIn.
Subscribe to receive updates from REPROCELL
Stem cell and drug discovery scientists around the world are using REPROCELL’s services and products in their preclinical and clinical research.